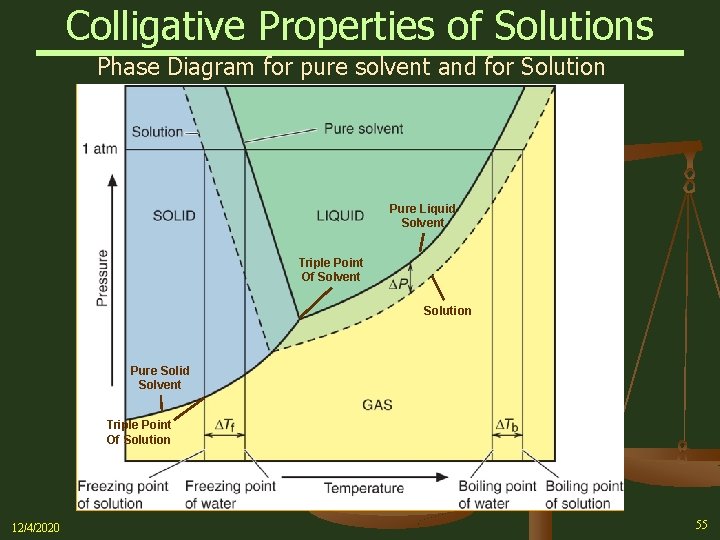
41 phase diagram of pure solvent and solution
Mixed Solvent systems - Phasediagram In all the depicted phase diagrams, the markers represent experimental data while the solubility equilibrium lines were calculated with the Extended UNIQUAC model. Notice that the temperatures vary between the diagrams. In all the graphs below, the focus is on the lower part of the diagrams. The apex with pure K 2 SO 4 is therefore not shown. label the phase diagram of pure solvent and a solution Label the phase ... label the phase diagram of pure solvent and a solution Label the phase diagram of pure solvent and a solution. Einen adventskalender mit rentier-motiven basteln Scherenschnitt schattenbilder Check out this awesome 'deer+shirt' design on @teepublic! Sandy lohß Hirsch berge als wandtattoo natur aufkleber deko Wandtattoo hirsch berge wanddeko ...
PDF Chapter 9: Phase Diagrams - University of Washington Phase Equilibria: Solubility Limit 65 Sucrose/Water Phase Diagram Pure Sugar emperature (°C) 0 20 40 60 80 100 C o =Composition (wt% sugar) L (liquid solution i.e., syrup) Solubility Limit L (liquid) + S (solid 20 sugar) 4 0 6 0 8 0 10 0 Pure ater Question: What is the solubility limit at 20C? Answer: 65wt% sugar. If Co < 65wt% sugar: syrup If ...

Phase diagram of pure solvent and solution
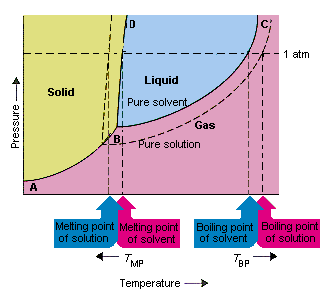
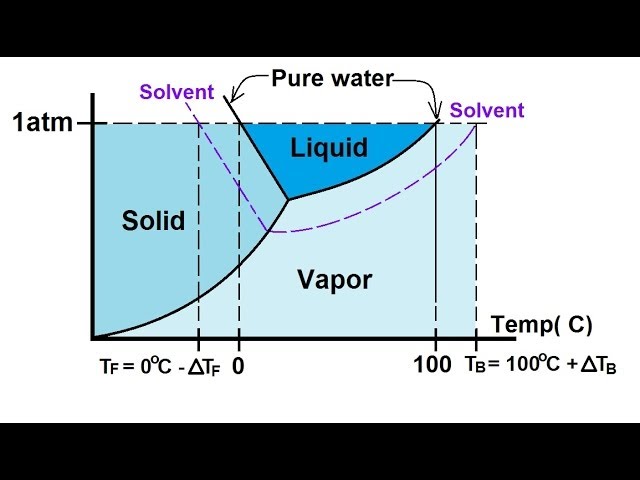
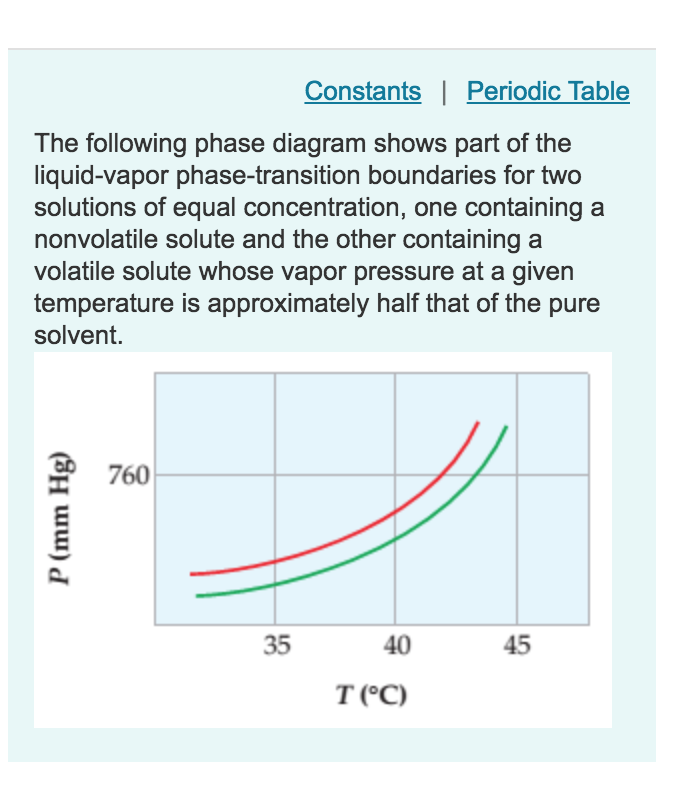
The figure shows two phase diagrams, one for a pure ... - Socratic.org Similarly, the normal boiling point of the pure substance at constant pressure is indicated by C, and the raised normal boiling point of the solution with the pure liquid as the solvent (again, due to addition of nonvolatile solute to the pure liquid substance) is indicated by D, since T ↑ rightwards. ΔT b = T b − T * b = iKbm Liquid/Solid Phase Diagram - an overview | ScienceDirect Topics (a) Construct the binary (solid + liquid) phase diagram and label all the regions. (b) Use the (T, x 2) data near x 2 = 0 and x 2 = 1 to calculate Δ fus H m for both components.Assume this Δ fus H m is constant with temperature and calculate the ideal freezing point at every 0.1 mole fraction unit. Plot these results on the diagram constructed in part (a) to compare the actual freezing ... The important differences between the phase diagram of a pure solvent ... The important differences between the phase diagram of a pure solvent and that of the solution of that solvent are to be determined. Concept introduction: The phase diagram is a combination of liquid-gas, solid-liquid, and solid-gas curves and determines the effect of temperature and pressure on the phase of a substance.
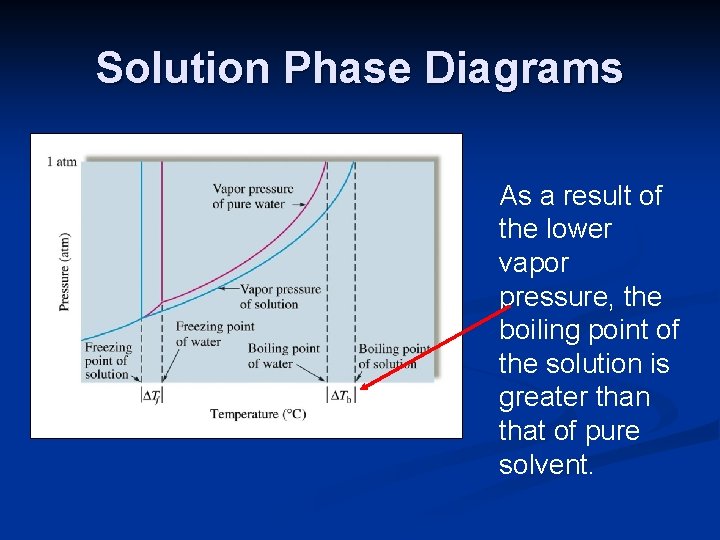
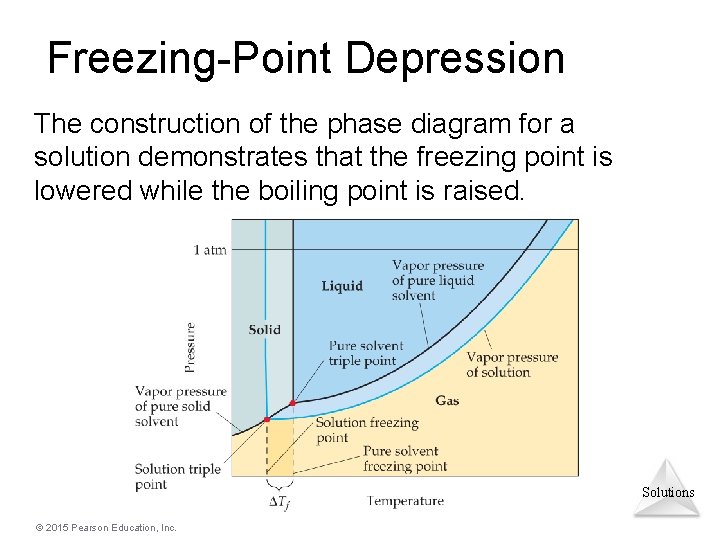
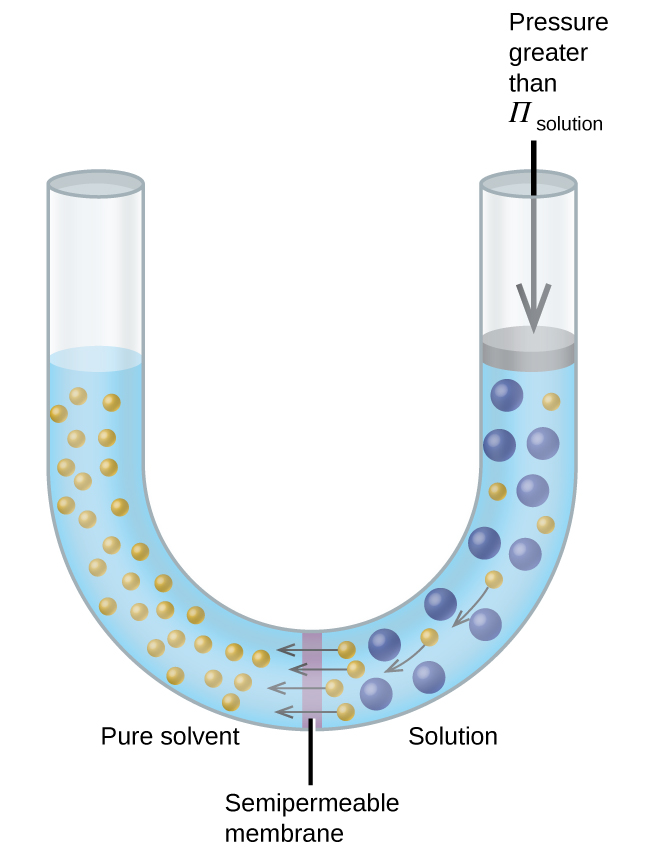
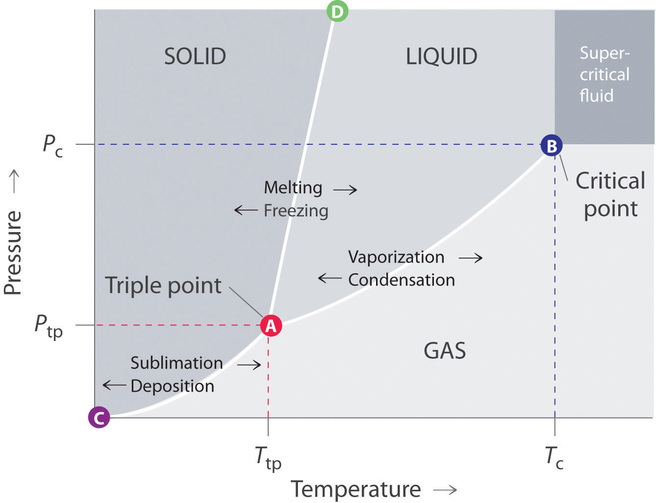
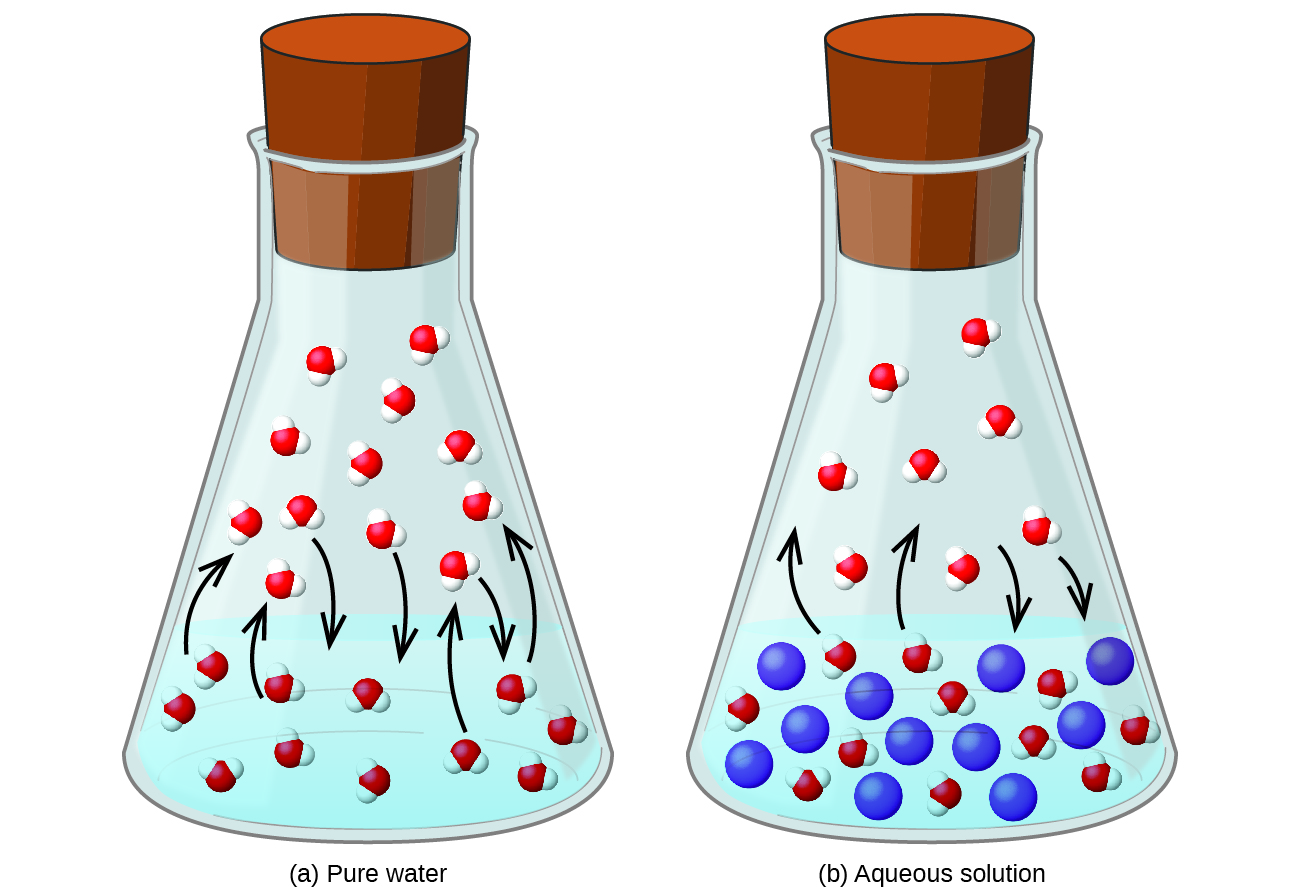
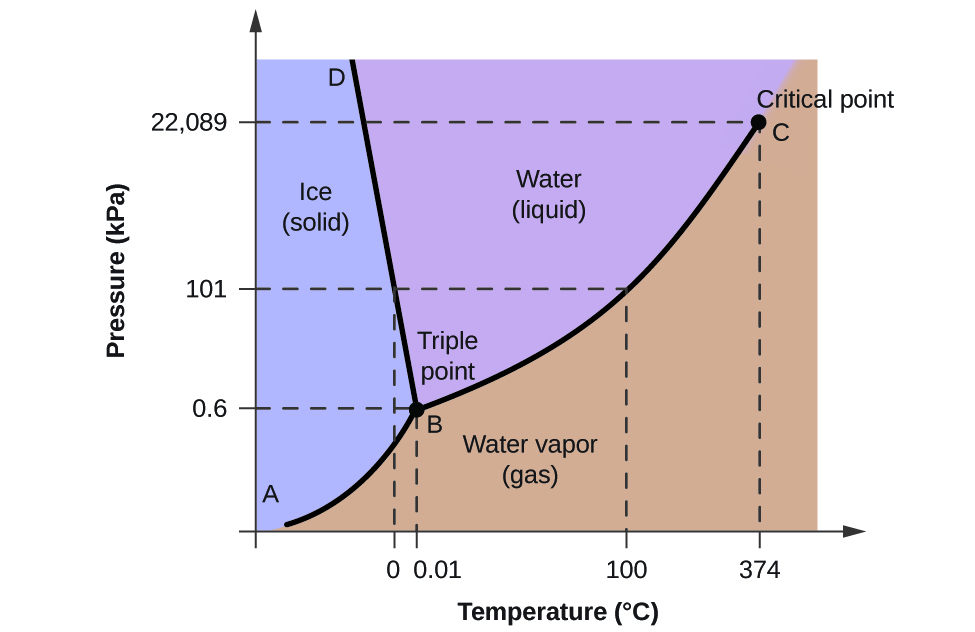
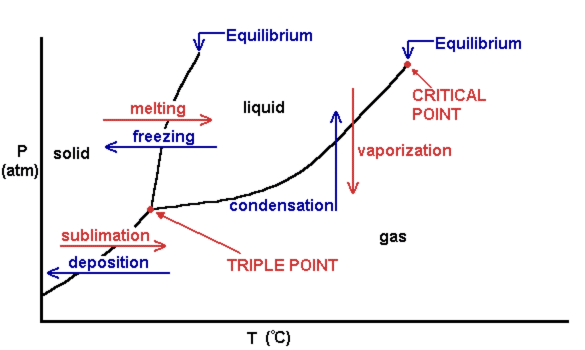
Phase diagram of pure solvent and solution. PHASE DIAGRAMS OF PURE SUBSTANCES - chemguide A phase diagram lets you work out exactly what phases are present at any given temperature and pressure. In the cases we'll be looking at on this page, the phases will simply be the solid, liquid or vapour (gas) states of a pure substance. This is the phase diagram for a typical pure substance. PDF Phase Diagrams Phase Diagrams Revised: 1/27/16 7 8. Slowly open the needle valve. Once the pressure within the system has equalized with atmospheric pressure, turn off the vacuum pump. 9. Melt the tert-butanol completely by suspending the 50 mL filter flask in a warm water bath. 10. What happens to the solute when a solution freezes? I came across a situation in which my chemistry textbook gives phase diagram of a pure solvent and a solution with the same solvent.I am considering 2 different , say beakers, one containing some amount of pure solvent , and the other contains a solution formed by a non volatile solute and the same amount of solvent. Phase Diagrams - Chemistry - University of Hawaiʻi Use phase diagrams to identify stable phases at given temperatures and pressures, and to describe phase transitions resulting from changes in these properties. In the previous module, the variation of a liquid's equilibrium vapor pressure with temperature was described. Considering the definition of boiling point, plots of vapor pressure ...
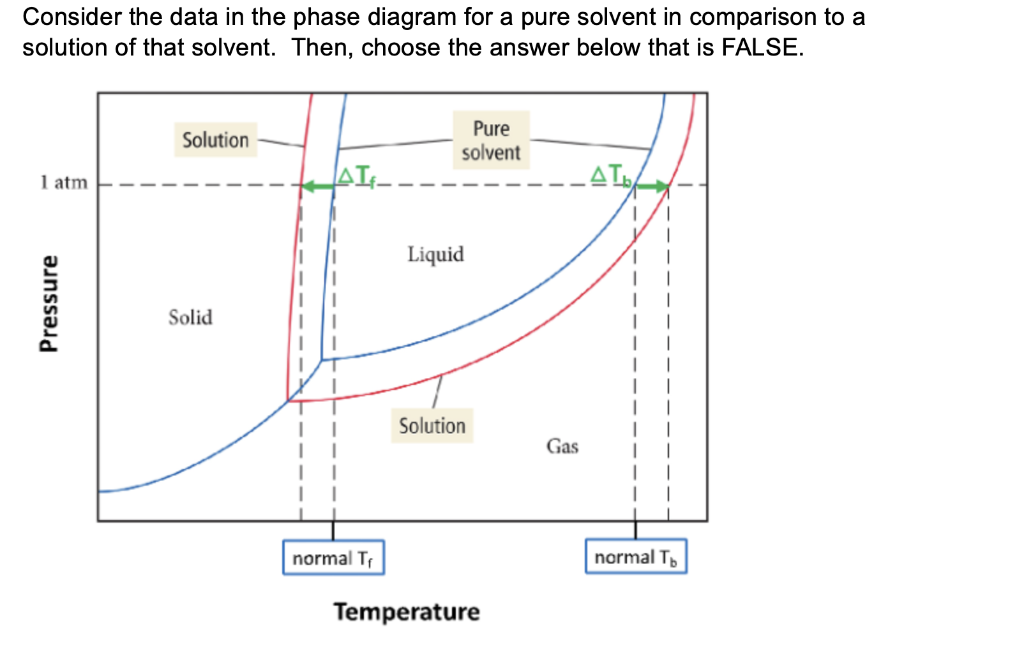
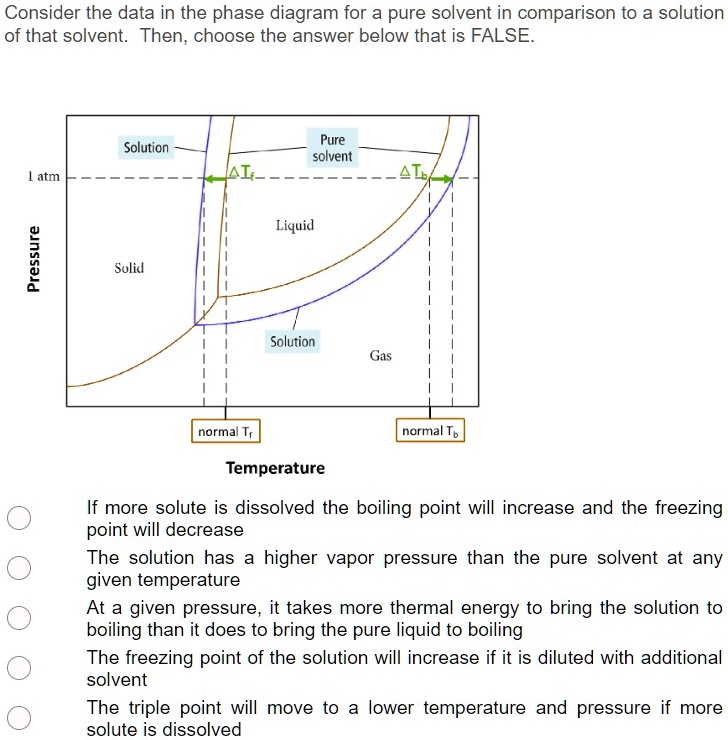
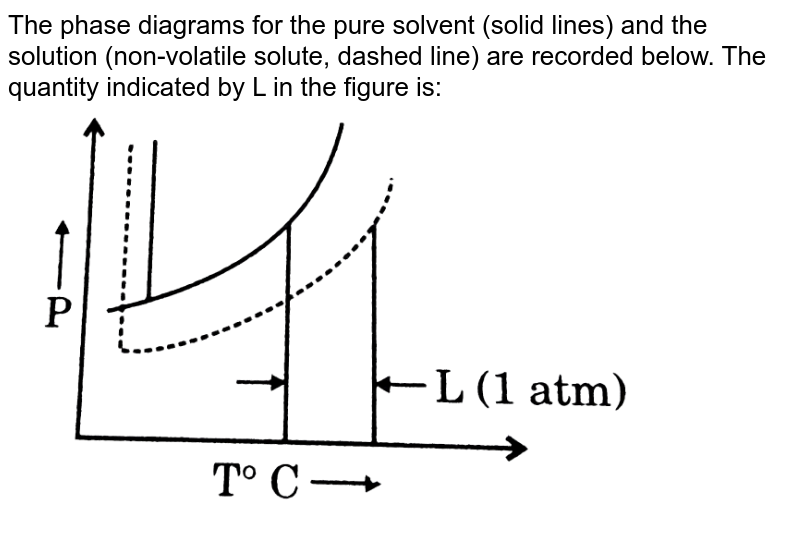
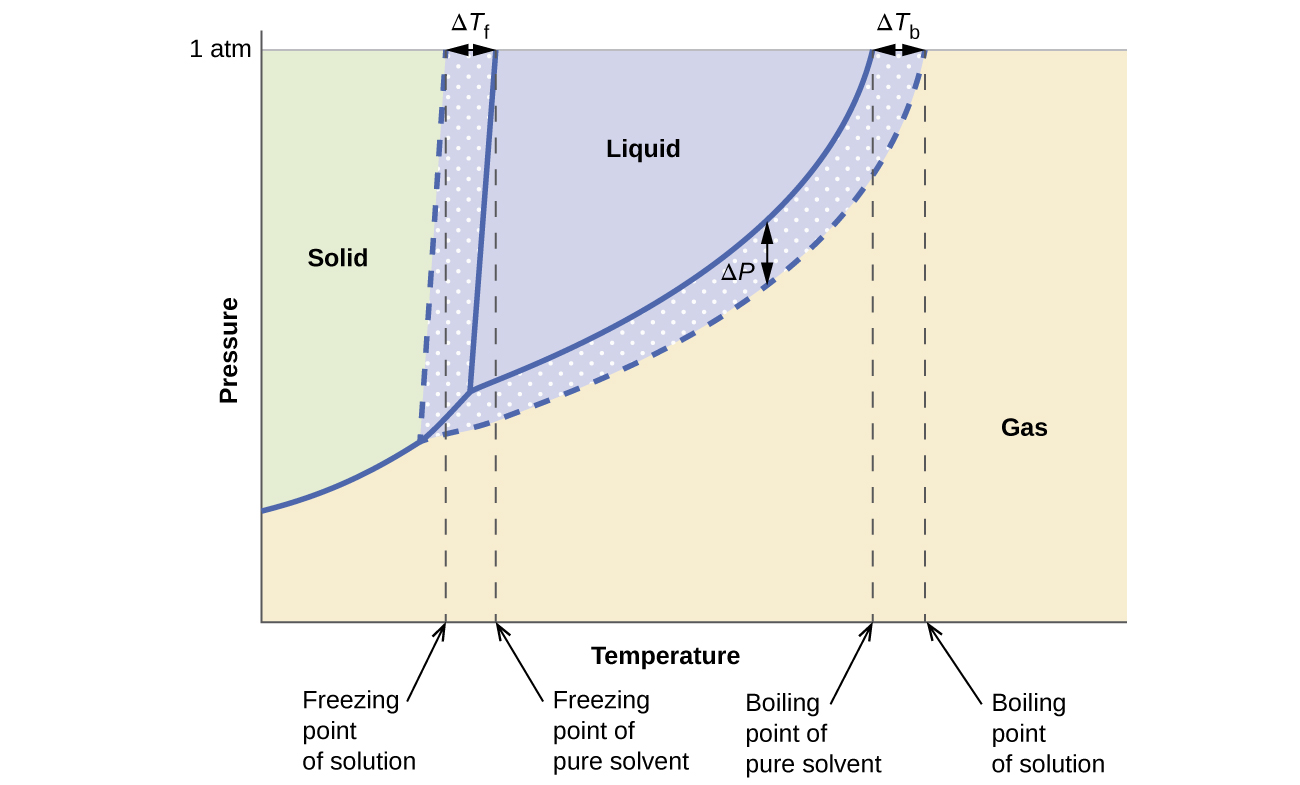
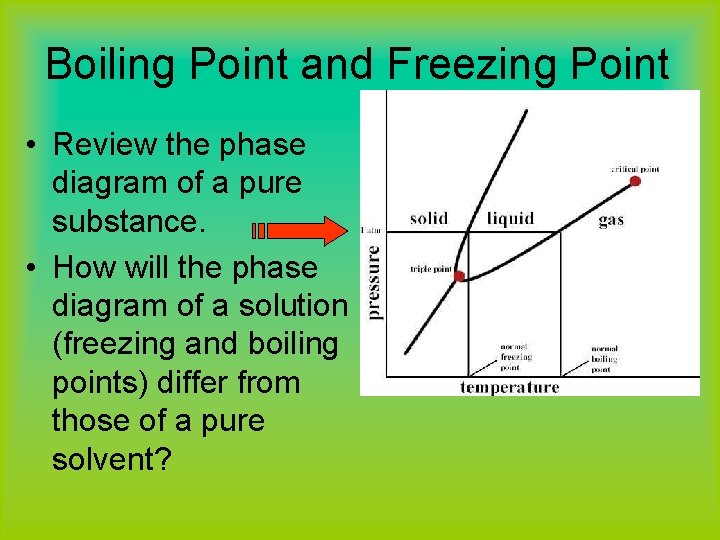
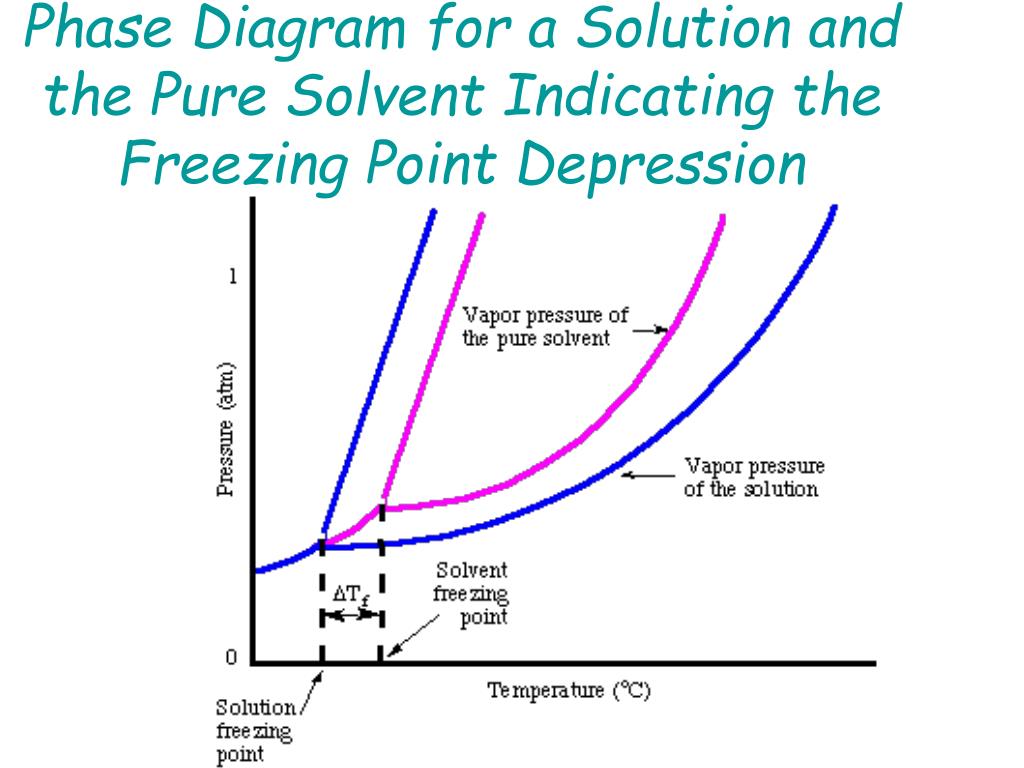
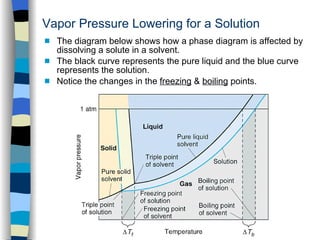
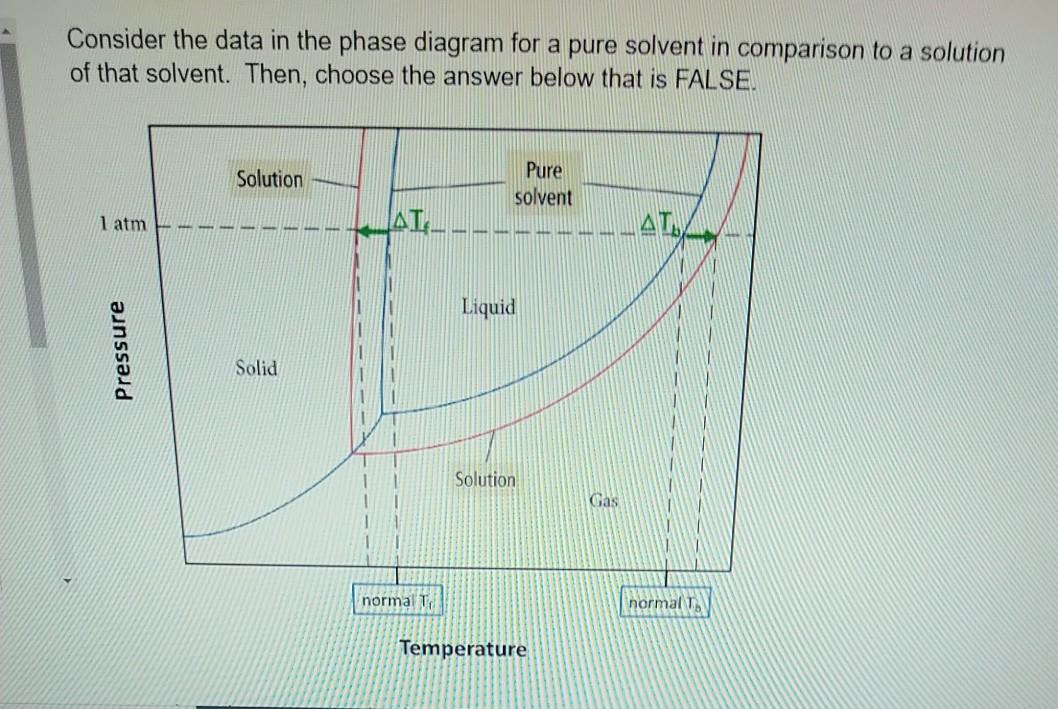
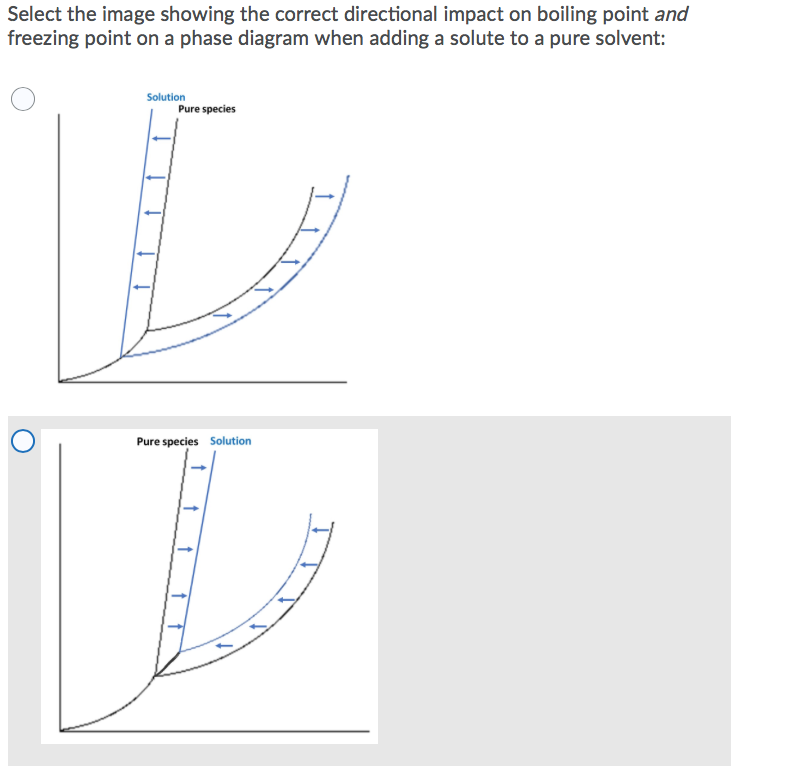
What are the most important differences between the phase di | Quizlet Two differences we can see on phase diagrams of a pure solvent and a solution are changes in phase transition temperatures \\textbf{phase transition temperatures} phase transition temperatures.Solution will always have a higher boiling point \\textbf{higher boiling point} higher boiling point and lower freezing point \\textbf{lower freezing point} lower freezing point (check pages 501, 502). Solved The phase diagrams for a pure solvent and the solvent - Chegg Transcribed image text: The phase diagrams for a pure solvent and the solvent in a solution are shown. Identify the normal freezing (fpsolv) and boiling (bpsolv) points for the pure solvent and the normal freezing (fpsoln) and boiling (bpsoln) points of the solution at 1 atm. Assume the solute is nonvolatile and that the solid that freezes from solution is pure solvent. 1 atm Liquid Solid ... The phase diagrams for the pure solvent (solid lines) and the solu | Filo The phase diagrams for the pure solvent (solid lines) and the solution (non-volatile solute, dashed line) are recorded below : The quantity indicated by L in the figure is : A Δ p Draw a phase diagram showing how the phase boundaries differ for a pure ... The phase boundaries are shown for both solvent and solution. The liquid gas boundary for a solution is at a higher temperature than that for the pure solvent. This indicates the elevation in boiling point. The liquid solid boundary is lower for the solution indicating the depression in freezing point.
SOLVED:The phase diagrams for = pure solvent and the solvent solution ... The phase diagrams for = pure solvent and the solvent solution are shown. Identify the normal freezing ((pwolv) and boiling (bpsolv) points for the pure solvent and the normal freezing (fpsoln) and boiling (bpsoln) points of the solution at atm_ Assume the solute nonvolatile and that the solid that freezes from solution pure solvent. atm Liquid 1 L Solid Answcr Bonk bpan Inaeln Gas Temperature pal Binary Solid-Liquid Phase Diagram | Chem Lab For more information on binary solid-liquid phase diagrams and the derivation of these equations, please see the references. 1-4 . From your data, generate the solid-liquid phase diagram for your system and determine Δ fus H and the melting point for the two pure substances at 95% confidence. Determine the eutectic temperature and composition ... Answered: The phase diagrams for a pure solvent… | bartleby identify the normal freezing (fpsolv) and boiling (bpsolv) points for the pure solvent and the normal freezing (fpsoln) and boiling (bpgoln) points of the solution at 1 atm. assume the solute is nonvolatile and that the solid that freezes from solution is pure solvent. 1 atm liquid solid answer bank fpsolv bpsolv fpsoln bpsoln gas temperature … PDF phase diagram solvent solution - justonly.com phase diagram solvent solution Effect of Solute on Phase Diagram of Water SOLIDLIQUID GAS Pure solvent Freezing point of solution Freezing point of water Boiling point of water Boiling point of solution 1 atm Pressure ΔTfΔTb ΔP Temperature
Solved The phase diagrams for a pure solvent and the solvent | Chegg.com Transcribed image text: The phase diagrams for a pure solvent and the solvent in a solution are shown. Identify the normal freezing (fp_solv) and boiling (bp_solv) points for the pure solvent and the normal freezing (fp_soln) and boiling (bp_soln) points of the solution at 1 atm. Assume the solute is nonvolatile and that the solid that freezes from solution is pure solvent.
PDF Phase Diagrams, Solid Solutions, Phase Transformations Phase Diagrams: composition of phases At TA= 1320°C: Only Liquid (L) present CL= C0 ( = 35 wt% Ni) At TB= 1250°C: Both and L present At TD= 1190°C: Only Solid ( ) present C = C0( = 35 wt% Ni) C L = C liquidus ( = 32 wt% Ni) C = C solidus ( = 43 wt% Ni) 18 • Rule 3:If we know T and Co, then we know: --the amount of each phase (given in wt%).
The phase diagrams for the pure solvent (solid lines) and the solution ... asked Dec 24, 2021 in Chemistry by SupriyaRani (24.2k points) The phase diagrams for the pure solvent (solid lines) and the solution (non-volatile solute, dashed line) are recorded below. The quantity indicated by L in the figure is: A. Δp Δ p B. ΔT f Δ T f C. K bm K b m D. K f m K f m class-12 graphical-interpretation
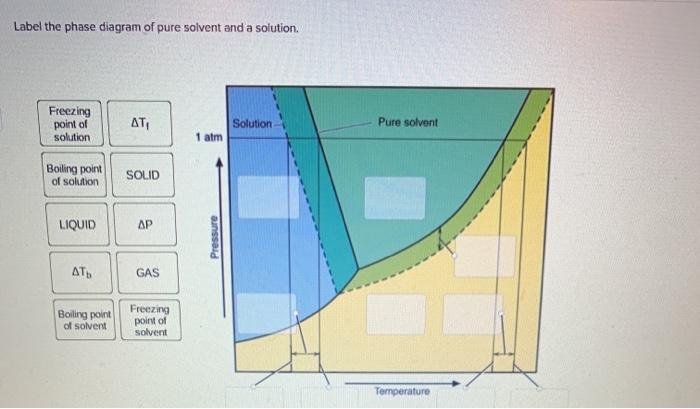
Solved Label the phase diagram of pure solvent and a - Chegg Experts are tested by Chegg as specialists in their subject area. We review their content and use your feedback to keep the quality high. Transcribed image text: Label the phase diagram of pure solvent and a solution Freezing point of solution GAS Solution Pure solvent 1 atm Boiling pointFreezing point of of solvent solvent ??? 11 SOLID AT AP ...
Solved: What are the most important differences between the phase ... Solutions for Chapter 13 Problem 60P: What are the most important differences between the phase diagram of a pure solvent and the phase diagram of a solution of that solvent? ...
Percentage recoveries of pure organic solvents after different mixing ... Download scientific diagram | Percentage recoveries of pure organic solvents after different mixing times with the aqueous phase. Data are mean ± range, n = 3 from publication: Exergy-based ...
Chapter 8 Phase Diagrams - Central Michigan University Example 8.2 Interpreting a liquid-liquid phase diagram . A mixture of 50 g of hexane (0.59 mol) and 50 g nitobenzene (0.41 mol) was prepared at 290 K. ... Pure solid B begins to come out of solution and the remaining liquid becomes richer in A. (2) a 2 ® a 3. More of the solid forms, and the relative amounts of the solid and liquid (which are ...
Phase diagram of pure solvent and solution. | Download Scientific Diagram Phase diagram of pure solvent and solution. Source publication Design and Evaluation of Multiple Effect Evaporator Systems According to the Steam Ejector Position Article Full-text available Jan...
Solved Label the phase diagram of pure solvent and a - Chegg Science. Chemistry. Chemistry questions and answers. Label the phase diagram of pure solvent and a solution.
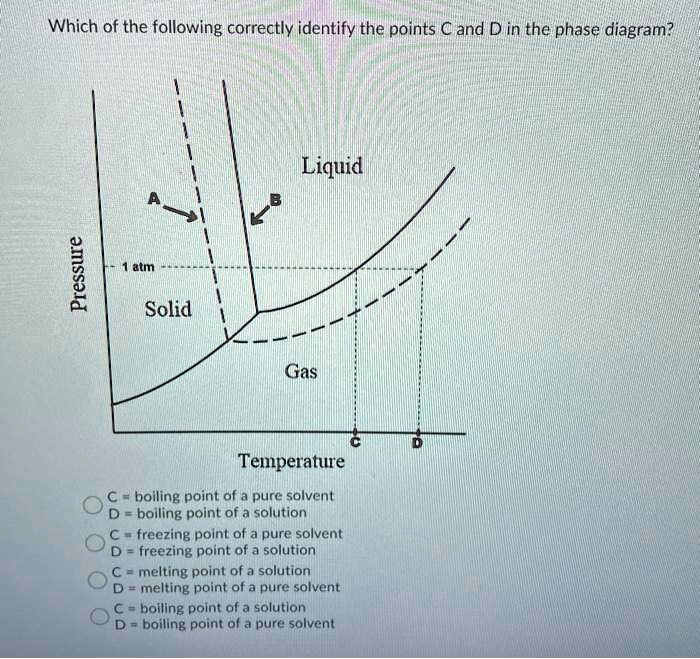
The phase diagram for solvent and solutions is shown in the figure ... The phase diagram for solvent and solutions is shown in the figure. What represents the normal boiling point of the solution? A A B B C C D D Hard Solution Verified by Toppr Correct option is D) The normal boiling point of the solution is that temperature at which vapour pressure of solution equals to 1 atm.
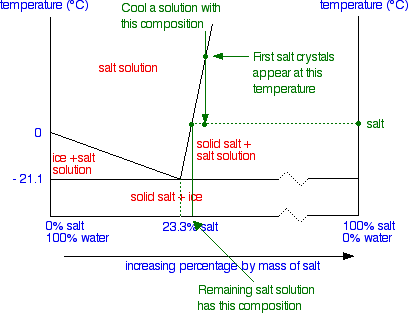
solid-liquid phase diagrams: salt solution - chemguide We'll take a solution containing 100 g of potassium nitrate and 100 g of water. Now let the solution cool. At all temperatures above that marked on the graph (about 57°C), 100 g of water will dissolve more than 100 g of potassium nitrate. All the potassium nitrate will stay in solution. At 57°C, you hit the solubility curve.
The phase diagrams for the pure solvent (solid lines) and the solution ... The phase diagrams for the pure solvent (solid lines) and the solution (non-volatile solute, dashed line) are recorded below: The quantity indicated by `L` i...
The important differences between the phase diagram of a pure solvent ... The important differences between the phase diagram of a pure solvent and that of the solution of that solvent are to be determined. Concept introduction: The phase diagram is a combination of liquid-gas, solid-liquid, and solid-gas curves and determines the effect of temperature and pressure on the phase of a substance.
Liquid/Solid Phase Diagram - an overview | ScienceDirect Topics (a) Construct the binary (solid + liquid) phase diagram and label all the regions. (b) Use the (T, x 2) data near x 2 = 0 and x 2 = 1 to calculate Δ fus H m for both components.Assume this Δ fus H m is constant with temperature and calculate the ideal freezing point at every 0.1 mole fraction unit. Plot these results on the diagram constructed in part (a) to compare the actual freezing ...
The figure shows two phase diagrams, one for a pure ... - Socratic.org Similarly, the normal boiling point of the pure substance at constant pressure is indicated by C, and the raised normal boiling point of the solution with the pure liquid as the solvent (again, due to addition of nonvolatile solute to the pure liquid substance) is indicated by D, since T ↑ rightwards. ΔT b = T b − T * b = iKbm














Post a Comment for "41 phase diagram of pure solvent and solution"