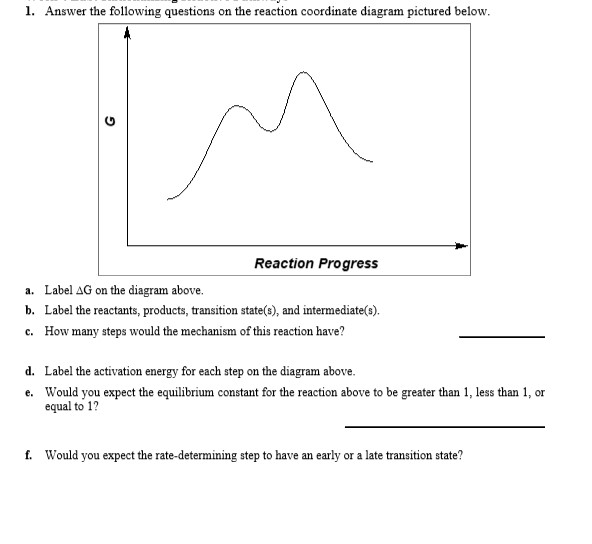
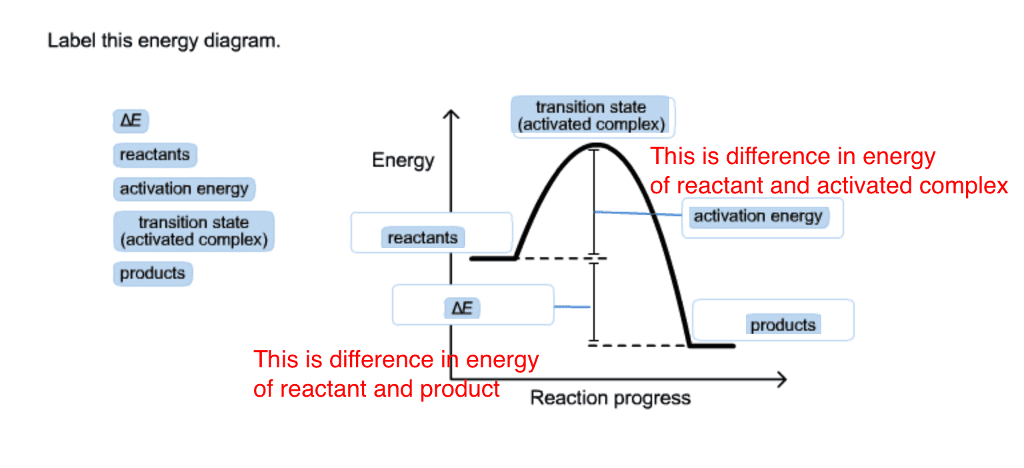
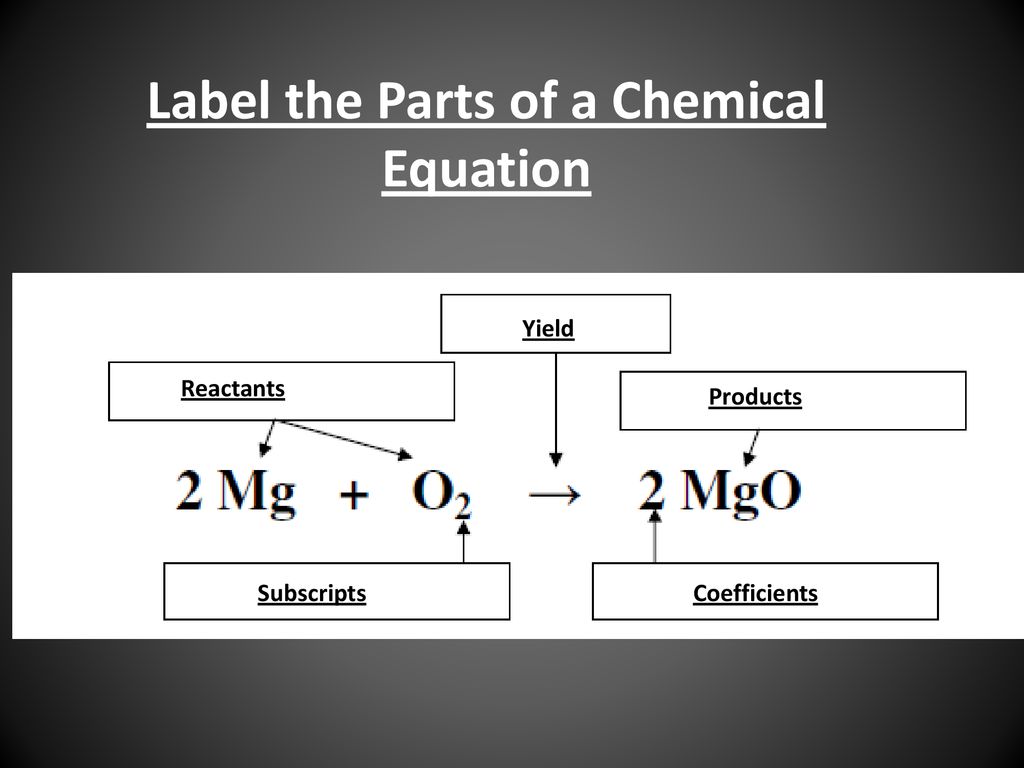
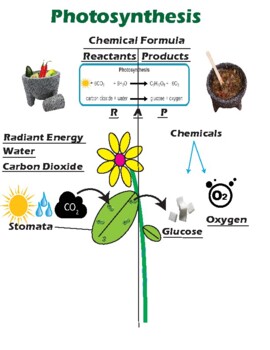
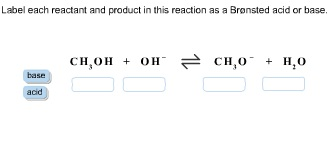
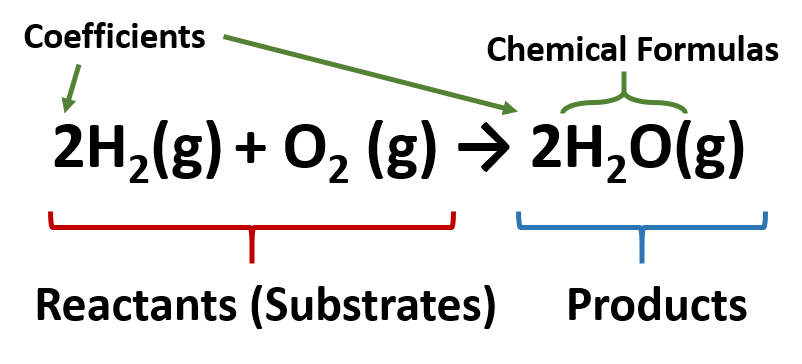
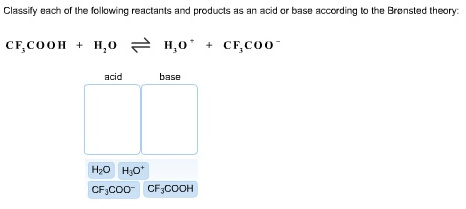
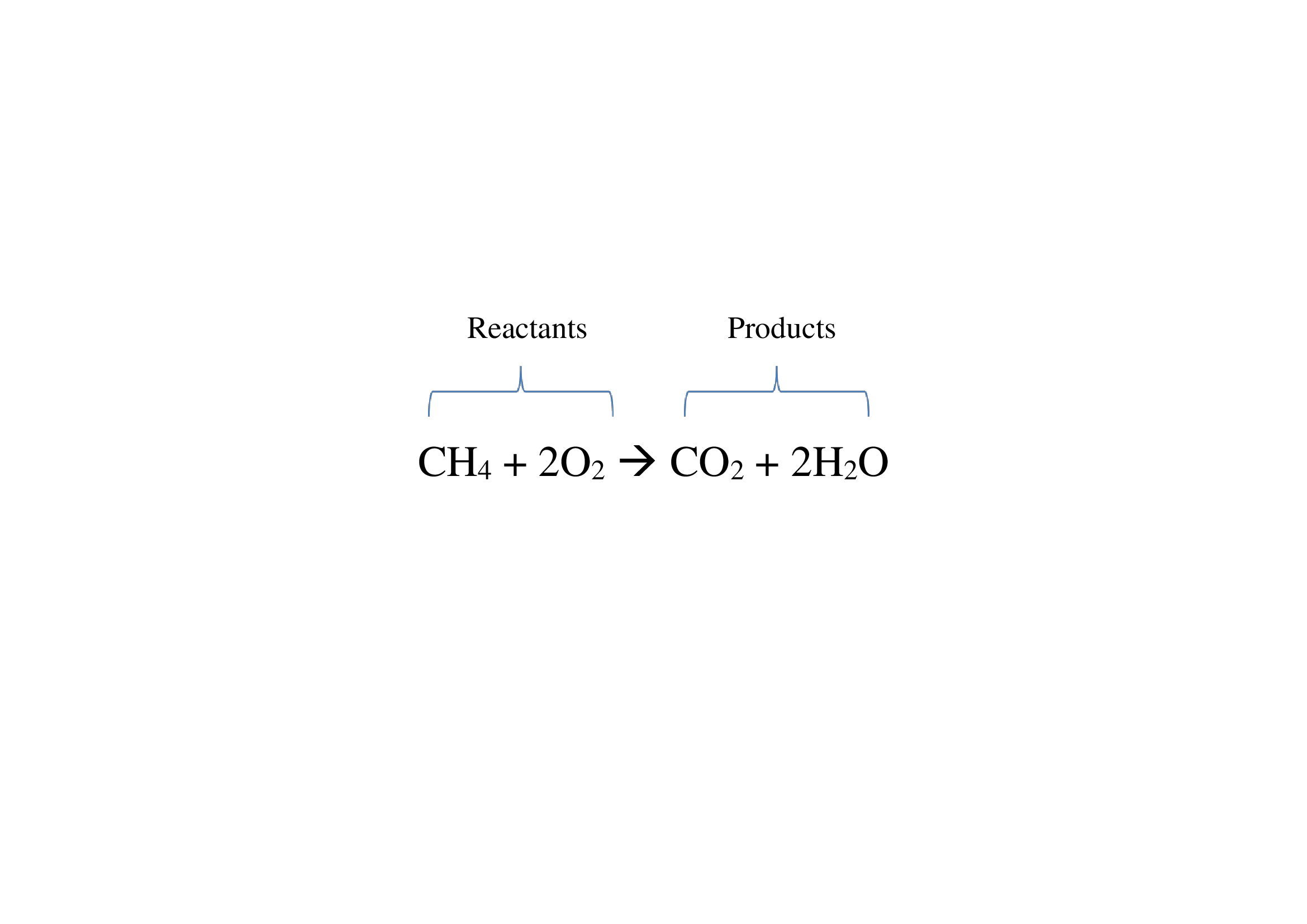
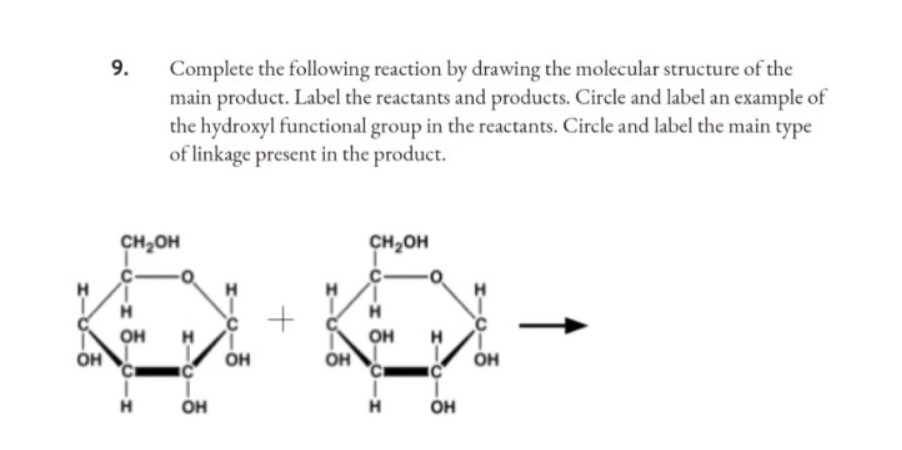
43 label the reactants and products
Rearrangement - Michigan State University In these two examples, diastereomeric reactants lead preferentially to diastereomeric intermediates, even though the anisyl group has a much greater migratory aptitude than phenyl. Electron pair donation by the hydroxyl substituent then acts to open the three-membered ring of these intermediates, yielding the ketone products. 4. Anchimeric ... Solving Hardy Weinberg Problems - YouTube Paul Andersen shows you how to solve simple Hardy-Weinberg problems. He starts with a brief description of a gene pool and shows you how the formula is deri...
Chemistry Quizzes | Study.com Practice with the Factor-Label Method . View Quiz. Interpreting Chemical Equations . ... Chemical Reactions & Resulting Products . View Quiz. Characteristics of Atoms . ... Reactants in Chemistry .

Label the reactants and products
KMnO4 + HCl = KCl + MnCl2 + Cl2 + H2O - ChemicalAid Label each compound (reactant or product) in the equation with a variable to represent the unknown coefficients. a KMnO 4 + b HCl = c KCl + d MnCl 2 + f Cl 2 + g H 2 O. Create a System of Equations. Create an equation for each element (K, Mn, O, H, Cl) where each term represents the number of atoms of the element in each reactant or product. 2.5: Reaction Rate - Chemistry LibreTexts Jun 17, 2022 · The Reaction Rate for a given chemical reaction is the measure of the change in concentration of the reactants or the change in concentration of the products per unit time. The speed of a chemical reaction may be defined as the change in concentration of a substance divided by the time interval during which this change is observed: 15.2: The Equilibrium Constant (K) - Chemistry LibreTexts Aug 14, 2020 · Many reactions have equilibrium constants between 1000 and 0.001 (\(10^3 \ge K \ge 10^{-3}\)), neither very large nor very small. At equilibrium, these systems tend to contain significant amounts of both products and reactants, indicating that there is not a strong tendency to form either products from reactants or reactants from products.
Label the reactants and products. Welcome to CK-12 Foundation | CK-12 Foundation FlexBook Platform®, FlexBook®, FlexLet® and FlexCard™ are registered trademarks of CK-12 Foundation. 15.2: The Equilibrium Constant (K) - Chemistry LibreTexts Aug 14, 2020 · Many reactions have equilibrium constants between 1000 and 0.001 (\(10^3 \ge K \ge 10^{-3}\)), neither very large nor very small. At equilibrium, these systems tend to contain significant amounts of both products and reactants, indicating that there is not a strong tendency to form either products from reactants or reactants from products. 2.5: Reaction Rate - Chemistry LibreTexts Jun 17, 2022 · The Reaction Rate for a given chemical reaction is the measure of the change in concentration of the reactants or the change in concentration of the products per unit time. The speed of a chemical reaction may be defined as the change in concentration of a substance divided by the time interval during which this change is observed: KMnO4 + HCl = KCl + MnCl2 + Cl2 + H2O - ChemicalAid Label each compound (reactant or product) in the equation with a variable to represent the unknown coefficients. a KMnO 4 + b HCl = c KCl + d MnCl 2 + f Cl 2 + g H 2 O. Create a System of Equations. Create an equation for each element (K, Mn, O, H, Cl) where each term represents the number of atoms of the element in each reactant or product.

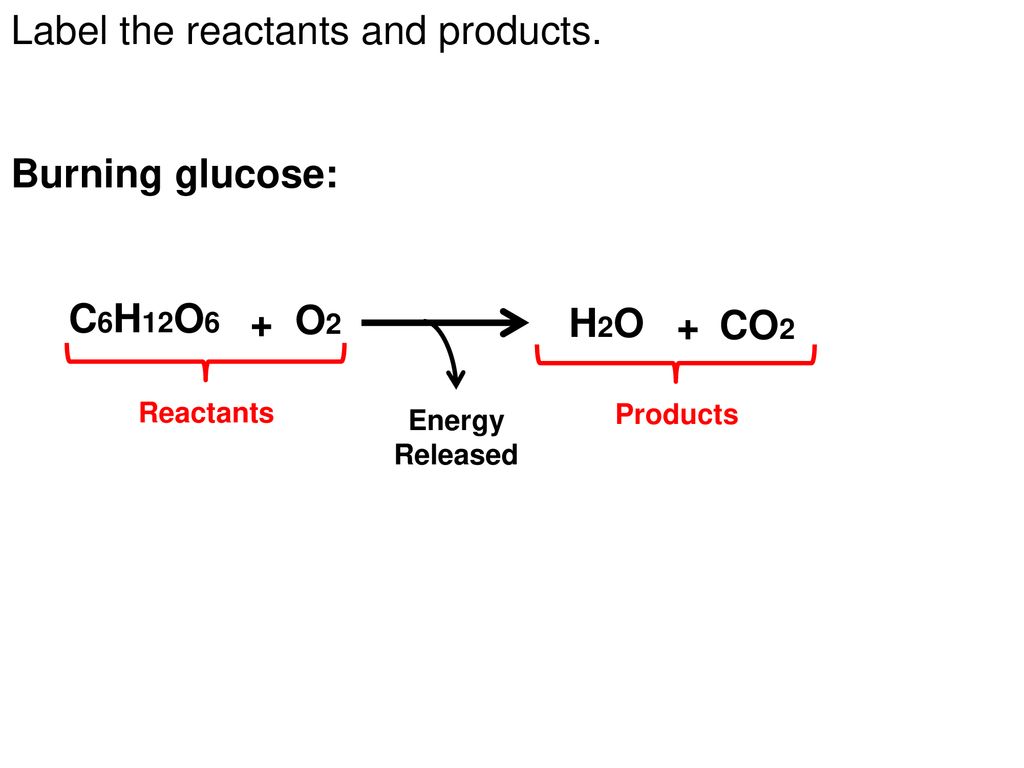
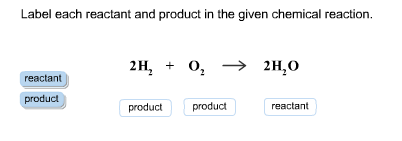
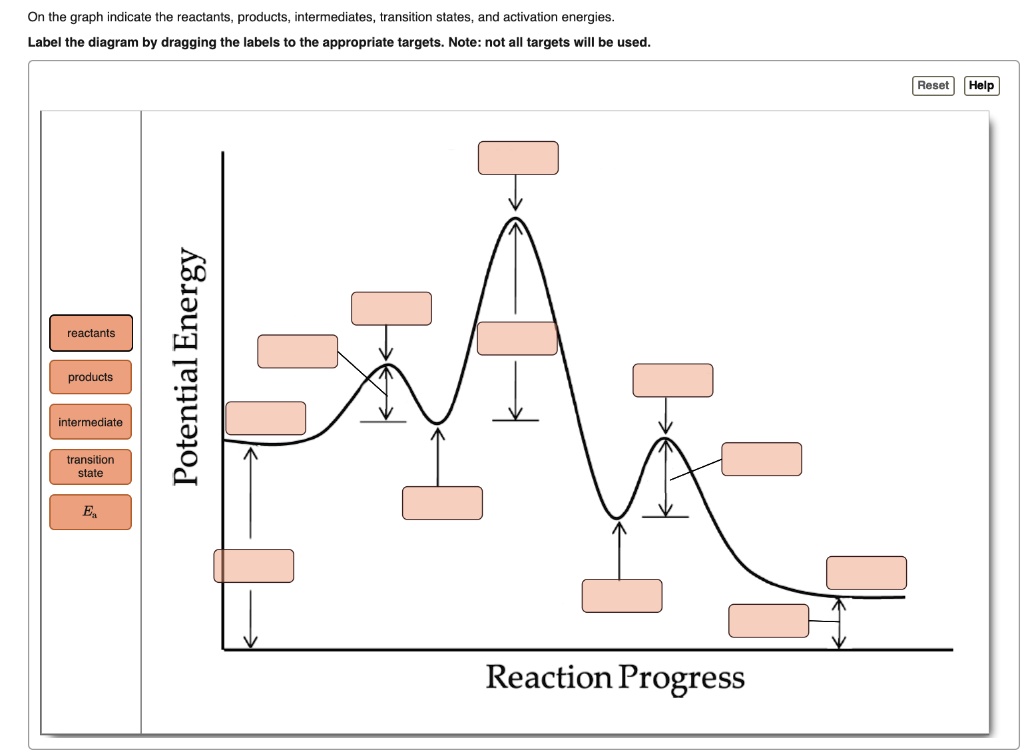



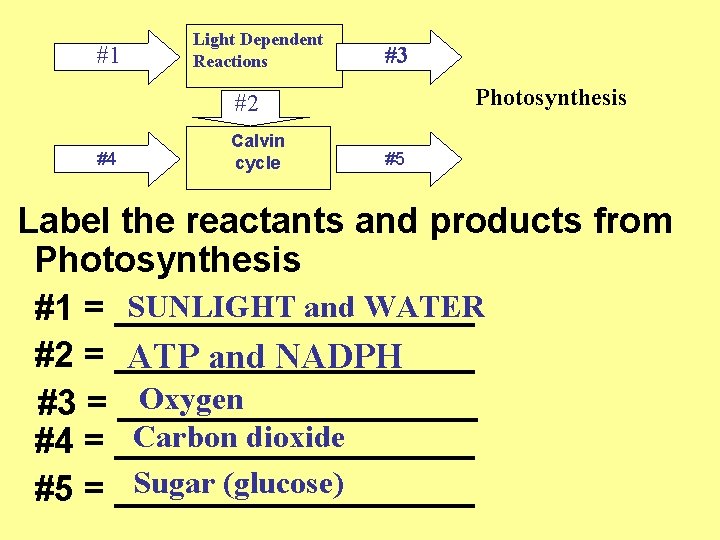









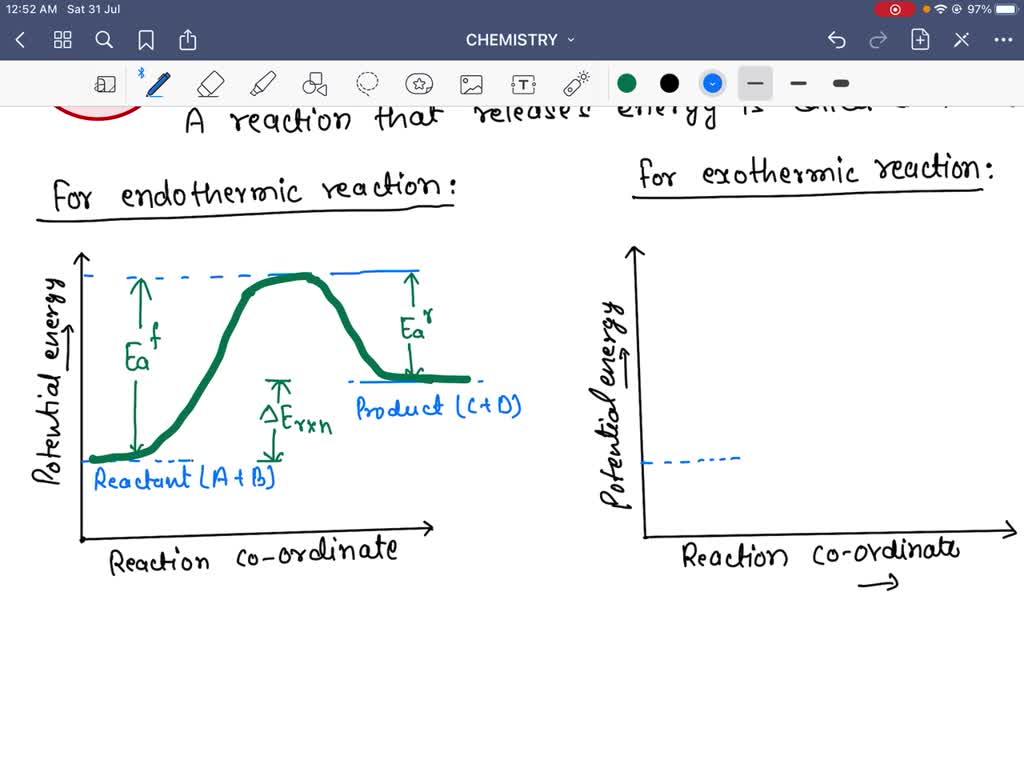
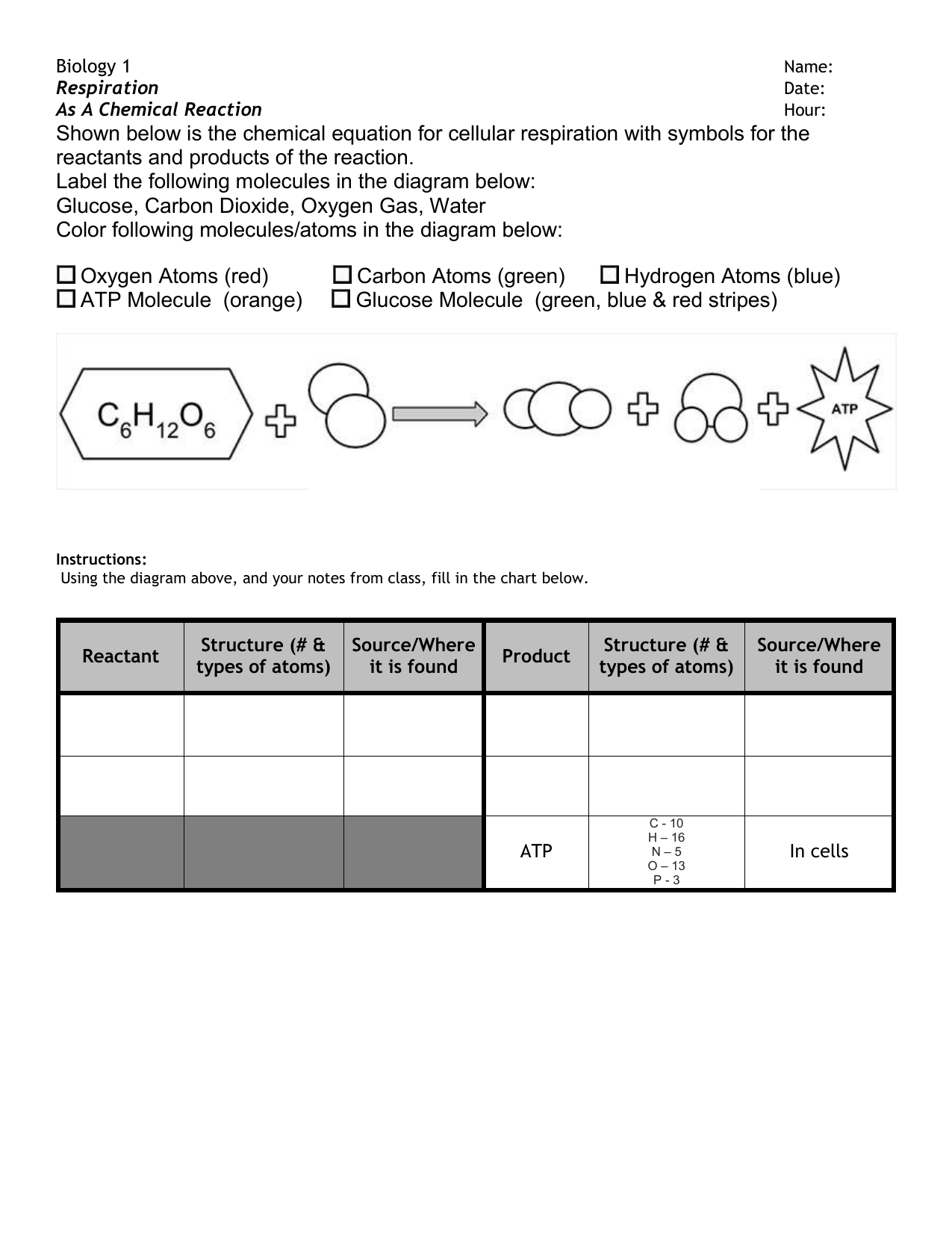





Post a Comment for "43 label the reactants and products"